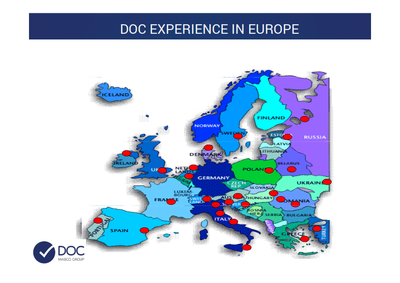
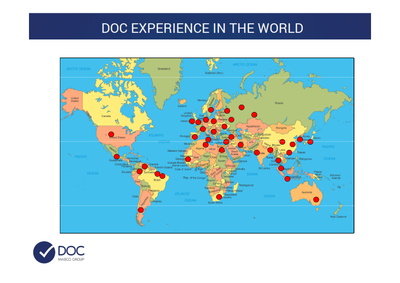
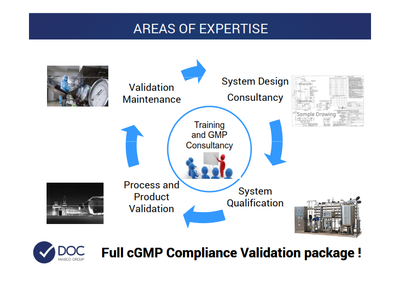
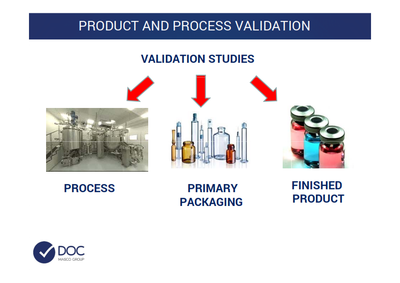
DOC - ltaly
www.docvalidation.it
Validation and consultancy services for cGMP compliance.
DOC is a more than 20 years old company and we work as validation company to provide, as final goal, a functional system fully validated.
Along with this service, we work also as a consultancy company: we can help the customers to write down the URS, we can perform a QRM Quality Risk management, we can provide a Conceptual design support and potential vendors evaluation for those customers that are about to build up new facilities.
Beside the Validation work, as globally intended, we provide also a Product validation that means Sterility test, LAL test, Antimicrobial Effectivness Test, etc.
Whatever is related to Data Integrity, Quality Risk Assesment , the Product and Process validation, all the PQ part , it is something that we can provide.
DOC is specialized in:
- System design
- System qualification
- Process validation
- Product validation
- Validation maintenance
- Training
- Consultancy
www.docvalidation.it
Validation and consultancy services for cGMP compliance.
DOC is a more than 20 years old company and we work as validation company to provide, as final goal, a functional system fully validated.
Along with this service, we work also as a consultancy company: we can help the customers to write down the URS, we can perform a QRM Quality Risk management, we can provide a Conceptual design support and potential vendors evaluation for those customers that are about to build up new facilities.
Beside the Validation work, as globally intended, we provide also a Product validation that means Sterility test, LAL test, Antimicrobial Effectivness Test, etc.
Whatever is related to Data Integrity, Quality Risk Assesment , the Product and Process validation, all the PQ part , it is something that we can provide.
DOC is specialized in:
- System design
- System qualification
- Process validation
- Product validation
- Validation maintenance
- Training
- Consultancy